RYPLAZIM cost and support
A Kedrion Nurse Educator will help you:
- Answer any questions you may have about RYPLAZIM and PLGD-1*
- Learn how to self-infuse if your doctor has approved you for at-home infusions
*Information from a Kedrion Nurse Educator should not replace your physician’s advice
A care coordinator from your specialty pharmacy will help you:
- Understand your insurance benefits and coverage options
- Find available financial assistance programs
- Manage product and infusion supply shipments (if your doctor has approved you for at-home infusions)
You may be eligible for financial assistance
The RYPLAZIM Co-Pay Assistance Program offers financial support to help with your deductible, co-pay, and/or coinsurance for RYPLAZIM.
To be eligible for co-pay assistance, you must:

Be commercially insured

Express a financial need

Have a health plan that permits members to participate in co-pay assistance programs
Restrictions: Not valid for prescriptions eligible for reimbursement by any federal or state healthcare programs, such as Medicare, Medicaid, Medigap, Veterans Affairs, Department of Defense, Tricare, or any other federal or state-funded programs.
Note: Nursing and supply costs that may be associated with the administration of the medication are not eligible for assistance through this program.
This program is subject to change or discontinuation by Kedrion at any time, for any reason, with or without prior notice.
See full terms and conditions here.
Talk to your healthcare provider or care coordinator about co-pay assistance
Learn moreFor more details, see the Plasminogen Deficiency Foundation's Insurance Approval Toolkit
View the toolkit
Downloadable resources
Connect with the community
APPROVED USE
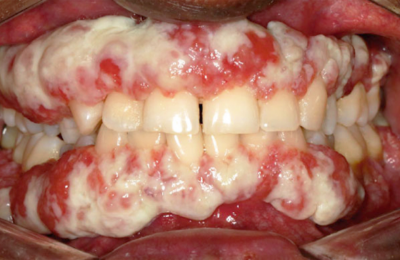
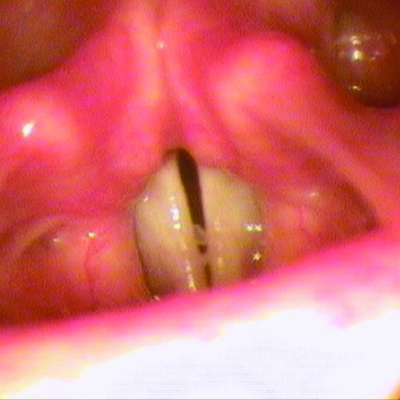
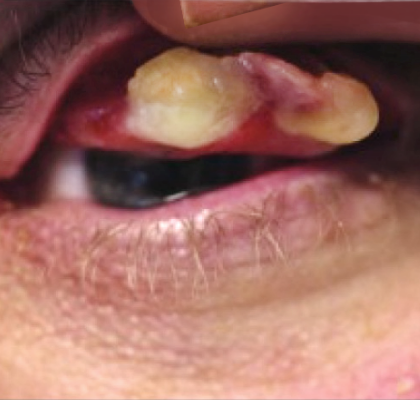
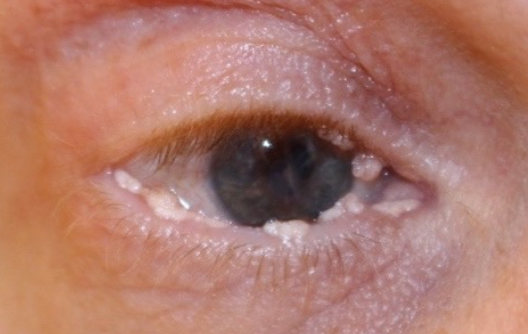
RYPLAZIM® (plasminogen, human-tvmh) is used for the treatment of patients with plasminogen deficiency type 1 (hypoplasminogenemia).
IMPORTANT SAFETY INFORMATION
Do not use RYPLAZIM if you are a patient with known hypersensitivity to plasminogen or other components of RYPLAZIM.
- Bleeding: Use of RYPLAZIM may lead to bleeding at disease-related lesion sites or worsen active bleeding not related to disease lesions. Discontinue RYPLAZIM if serious bleeding occurs. If you are treated at a healthcare provider’s office or center, your healthcare provider will need to monitor you during the infusion and for 4 hours afterward, especially if you have a predisposition to bleeding or are taking anticoagulants, antiplatelet drugs, or other medications that may interfere with normal coagulation. If you infuse RYPLAZIM at home, you or your caregiver must also monitor you during and for 4 hours after the infusion under the same circumstances.
- Tissue Sloughing: If you have mucosal lesions in your respiratory tract, especially the tracheobronchial tree, and are treated with RYPLAZIM, respiratory distress may occur due to tissue sloughing. Please monitor appropriately.
- Transmission of Infectious Agents: RYPLAZIM is made from human plasma and therefore carries a risk of transmitting infectious agents, e.g., viruses, the variant Creutzfeldt-Jakob disease (vCJD) agent, and theoretically, the Creutzfeldt-Jakob Disease (CJD) agent. You should consult with your healthcare provider if you have any questions or concerns.
- Hypersensitivity Reactions: Hypersensitivity reactions, including anaphylaxis, may occur with RYPLAZIM. Immediately report any of the following signs or symptoms of an allergic reaction to your healthcare provider: swelling of the throat, tightness of the chest, low blood pressure, rash, nausea, vomiting, a tingling or pricking sensation, restlessness, wheezing or shortness of breath.
- Neutralizing Antibodies: Antibodies neutralizing plasminogen (also known as inhibitors) which can make the product less effective or ineffective, may form with the use of RYPLAZIM, although they were not observed in clinical trials. If clinical efficacy is not maintained (e.g., development of new or recurrent lesions), your healthcare provider will determine low levels of plasminogen in your plasma.
- Laboratory Abnormalities: Due to the activity of RYPLAZIM, you may have elevated blood levels of D-dimer that may interfere with screening for venous thromboembolism (VTE) using D-dimer levels.
- Adverse Reactions: The most frequent (incidence ≥ 10%) adverse reactions in clinical trials were abdominal pain, bloating, nausea, fatigue, extremity pain, hemorrhage, constipation, dry mouth, headache, dizziness, arthralgia, and back pain.
You may report side effects to +1(855)-353-7466 and US_Medicalinfo@kedrion.com or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
For more details, please see full Prescribing Information and the Patient Information.
This site is intended for residents of the US only.