ABOUT RYPLAZIM
RYPLAZIM is a human plasminogen replacement therapy. It restores a key protein that people with PLGD-1 are missing.1
Replacing plasminogen helps the body break down lesions in the eyes, mouth, ears, and other parts of the body.1
Because plasminogen is gradually used and cleared from the body, RYPLAZIM is given on a regular schedule to help keep lesions from coming back.1,3
Leave lesions behind with RYPLAZIM
RYPLAZIM clinical success1,4*

ALL patients saw their
lesions improve*
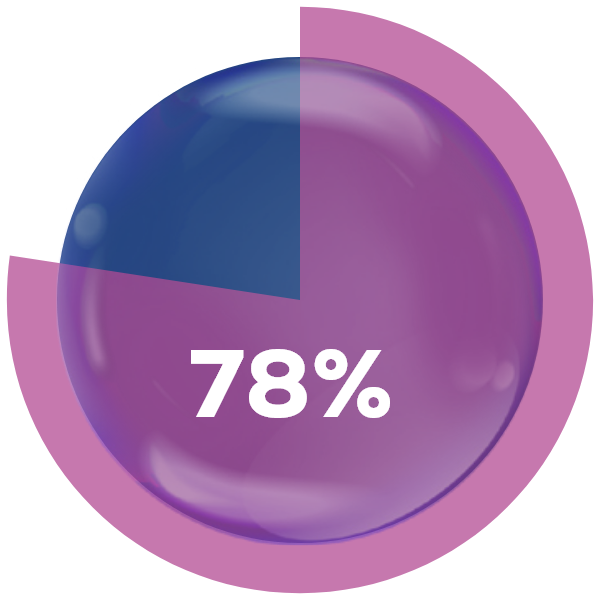 External
Externallesions
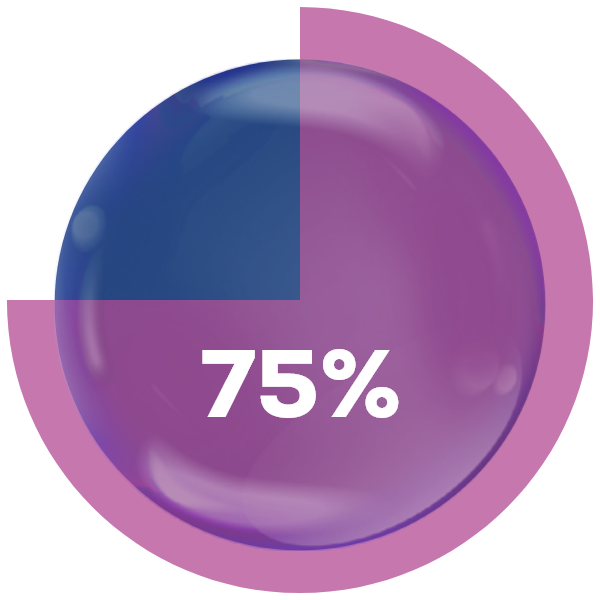 Internal
Internallesions
% of lesions fully
gone after week 48
Most patients saw their lesions
fully clear in only 8 weeks.

New or returning lesions in any patient through week 48
*Clinical success = a decrease in lesion number or size by at least half.
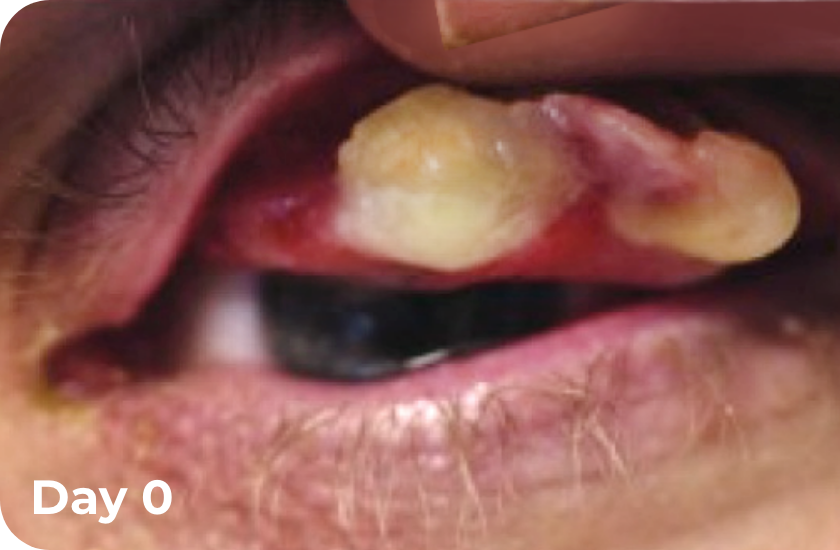
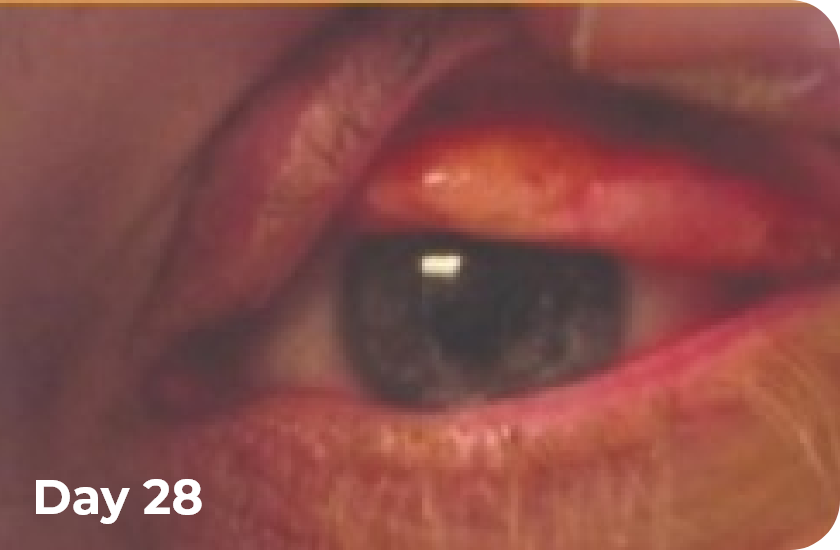
Eye lesions BEFORE and DURING RYPLAZIM5
Possible side effects of RYPLAZIM1
You are encouraged to report negative side effects of prescription drugs to the FDA. Call 1-800-FDA-1088 or visit www.fda.gov/medwatch.
In a separate study, 12 people treated with RYPLAZIM were
observed for ~5 years3

No new or returning lesions in people with uninterrupted treatment
RYPLAZIM was generally well tolerated

In some people, lesions returned after missing or lowering doses, then improved once recommended treatment was restarted
RYPLAZIM controls lesions as long as you stay on treatment3,4
If you've been diagnosed with PLGD-1, RYPLAZIM could be right for you. Talk to your doctor and connect with a nurse educator to learn more about RYPLAZIM.
Learn moreRYPLAZIM is carefully made with your safety in mind

Only healthy plasma donors are accepted at FDA-licensed centers.1,6

Every plasma donation is carefully checked for harmful viruses.1

Special filters and treatments are used to help remove and inactivate viruses during the creation of RYPLAZIM.1
APPROVED USE
RYPLAZIM® (plasminogen, human-tvmh) is used for the treatment of patients with plasminogen deficiency type 1 (hypoplasminogenemia).
IMPORTANT SAFETY INFORMATION
Do not use RYPLAZIM if you are a patient with known hypersensitivity to plasminogen or other components of RYPLAZIM.
- Bleeding: Use of RYPLAZIM may lead to bleeding at disease-related lesion sites or worsen active bleeding not related to disease lesions. Discontinue RYPLAZIM if serious bleeding occurs. If you are treated at a healthcare provider’s office or center, your healthcare provider will need to monitor you during the infusion and for 4 hours afterward, especially if you have a predisposition to bleeding or are taking anticoagulants, antiplatelet drugs, or other medications that may interfere with normal coagulation. If you infuse RYPLAZIM at home, you or your caregiver must also monitor you during and for 4 hours after the infusion under the same circumstances.
- Tissue Sloughing: If you have mucosal lesions in your respiratory tract, especially the tracheobronchial tree, and are treated with RYPLAZIM, respiratory distress may occur due to tissue sloughing. Please monitor appropriately.
- Transmission of Infectious Agents: RYPLAZIM is made from human plasma and therefore carries a risk of transmitting infectious agents, e.g., viruses, the variant Creutzfeldt-Jakob disease (vCJD) agent, and theoretically, the Creutzfeldt-Jakob Disease (CJD) agent. You should consult with your healthcare provider if you have any questions or concerns.
- Hypersensitivity Reactions: Hypersensitivity reactions, including anaphylaxis, may occur with RYPLAZIM. Immediately report any of the following signs or symptoms of an allergic reaction to your healthcare provider: swelling of the throat, tightness of the chest, low blood pressure, rash, nausea, vomiting, a tingling or pricking sensation, restlessness, wheezing or shortness of breath.
- Neutralizing Antibodies: Antibodies neutralizing plasminogen (also known as inhibitors) which can make the product less effective or ineffective, may form with the use of RYPLAZIM, although they were not observed in clinical trials. If clinical efficacy is not maintained (e.g., development of new or recurrent lesions), your healthcare provider will determine low levels of plasminogen in your plasma.
- Laboratory Abnormalities: Due to the activity of RYPLAZIM, you may have elevated blood levels of D-dimer that may interfere with screening for venous thromboembolism (VTE) using D-dimer levels.
- Adverse Reactions: The most frequent (incidence ≥ 10%) adverse reactions in clinical trials were abdominal pain, bloating, nausea, fatigue, extremity pain, hemorrhage, constipation, dry mouth, headache, dizziness, arthralgia, and back pain.
You may report side effects to +1(855)-353-7466 and US_Medicalinfo@kedrion.com or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
For more details, please see full Prescribing Information and the Patient Information.
This site is intended for residents of the US only.
References:
1. RYPLAZIM [prescribing information]. Kedrion Biopharma Inc. 2024.
2. FDA approves first treatment for patients with plasminogen deficiency, a rare genetic disorder. US Food & Drug Administration. Updated June 4, 2021. Accessed February 5, 2026. https://www.fda.gov/news-events/press-announcements/fda-approves-first-treatment-patients-plasminogen-deficiency-rare-genetic-disorder
3. Shapiro AD, McDaniel H, Decker RW, et al. Safety and efficacy of long-term treatment of type 1 plasminogen deficient patients with intravenous plasminogen replacement therapy. Haemophilia. 2025;31(3):477-484.
4. Shapiro AD, Nakar C, Parker JM, Thibaudeau K, Crea R, Sandset PM. Plasminogen, human-tvmh for the treatment of children and adults with plasminogen deficiency type 1. Haemophilia. 2023;29(6):1556-1564.
5. Data on file. Kedrion Biopharma Inc.
6. International Quality Plasma Program (IQPP). Plasma Protein Therapeutics Association. Accessed February 5, 2026. https://www.pptaglobal.org/material/international-quality-plasma-program-iqpp

